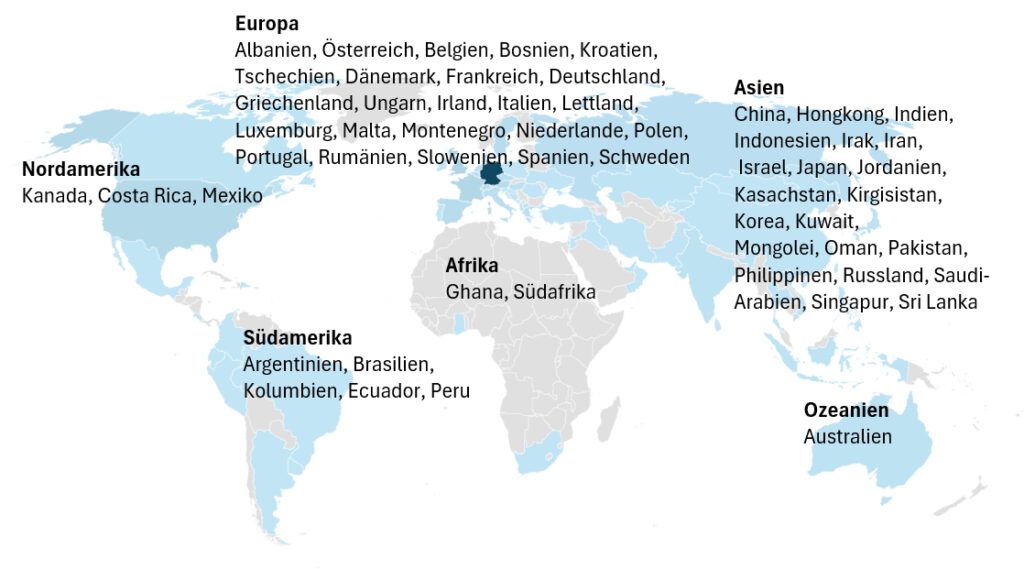
Wir auditieren weltweit
SCHOLZ Pharma GmbH was founded by Dr. Bianca Scholz in 2007 and offers business consulting and services in the field of pharmaceuticals (pharmaceutical / medical / healthcare). In addition to our core competencies of audit and inspections in the areas of GCP, GLP, GMP and GVP, our team at SCHOLZ Pharma GmbH also offer customized solutions in the field of pharmaceutical management, coaching, training and document management from development to market presence. SCHOLZ Pharma GmbH works closely together as a team in order to be able to offer you a comprehensive service in the field of GxP. Our experts for the respective areas of GxP support you individually or as a team to meet your specific requirements. In addition to our core competencies, SCHOLZ Pharma GmbH works closely with experts in the following guidelines.
Satisfied customers who are convinced of our services. We are proud of this and give our best every day.

Als Unternehmen ist es uns wichtig, Verantwortung nicht nur in unseren Projekten, sondern auch darüber hinaus zu übernehmen. Deshalb engagiert sich die ScholzPharma GmbH gezielt für gemeinnützige Initiativen, die Menschen in herausfordernden Lebenssituationen unterstützen – regional wie international.
Unser Engagement folgt dabei einem klaren Grundsatz:
Hilfe soll dort ankommen, wo sie gebraucht wird – respektvoll, transparent und ohne Erwartung einer Gegenleistung.
Mit einer Spende an den Hospiz Bergstraße e. V. unterstützen wir die ambulante Hospizarbeit in unserer Region. Der Verein begleitet schwerstkranke Menschen sowie deren Angehörige in einer besonders sensiblen Lebensphase – mit Zeit, Zuwendung und Menschlichkeit.
Die Arbeit des Hospizvereins ermöglicht es Betroffenen, auch am Lebensende in Würde, Selbstbestimmung und mit professioneller Begleitung zu leben. Dieses Engagement verdient unseren höchsten Respekt und unsere Unterstützung.
Neben gezielten Spenden ist gesellschaftliches Engagement für uns auch eine Frage der Haltung. Alle Mitarbeitenden der ScholzPharma GmbH sind Mitglied im Verein „Wir sind Bergstraße e. V.“ und bekennen sich damit aktiv zur Förderung regionaler Initiativen, Gemeinschaft und Zusammenhalt.
Der Verein Wir sind Bergstraße e. V. setzt sich seit seiner Gründung von Dr. Bianca Scholz und Annalena Homa für gemeinnützige und humanitäre Projekte ein. Entstanden ist das Engagement während der COVID‑19‑Pandemie, als in der Region Bergstraße kurzfristig ehrenamtliche Hilfe organisiert wurde, um Pflegeeinrichtungen und medizinisches Personal mit dringend benötigter Schutzausrüstung zu unterstützen.
In den folgenden Jahren entwickelte sich daraus ein dauerhaftes Netzwerk für konkrete Hilfe. Der Verein organisierte Spendenaktionen und Hilfstransporte und unterstützte insbesondere humanitäre Projekte in Krisengebieten. Ein aktueller Schwerpunkt lag dabei auf der Unterstützung der medizinischen Versorgung im Zusammenhang mit dem Krieg in der Ukraine – unter anderem durch die Koordination von Geld‑ und Sachspenden sowie logistische Hilfe in Zusammenarbeit mit regionalen Partnern.
Neben internationalen Hilfsprojekten engagiert sich der Verein auch für gemeinnützige Initiativen in der Region Bergstraße und stärkt damit gesellschaftlichen Zusammenhalt und bürgerschaftliches Engagement.
Dieses gemeinsame Engagement stärkt nicht nur die Region, sondern auch unser Miteinander als Team.



Unsere Spenden und Unterstützungen erfolgen nach internen Kriterien und ausschließlich an anerkannte, gemeinnützige Organisationen. Die Verwendung der Mittel ist nachvollziehbar dokumentiert. Eine Unterstützung erfolgt stets ohne wirtschaftliche oder sonstige Gegenleistung.
So möchten wir einen kleinen, aber bewussten Beitrag leisten – für Menschen, für Gemeinschaften und für eine solidarische Gesellschaft.